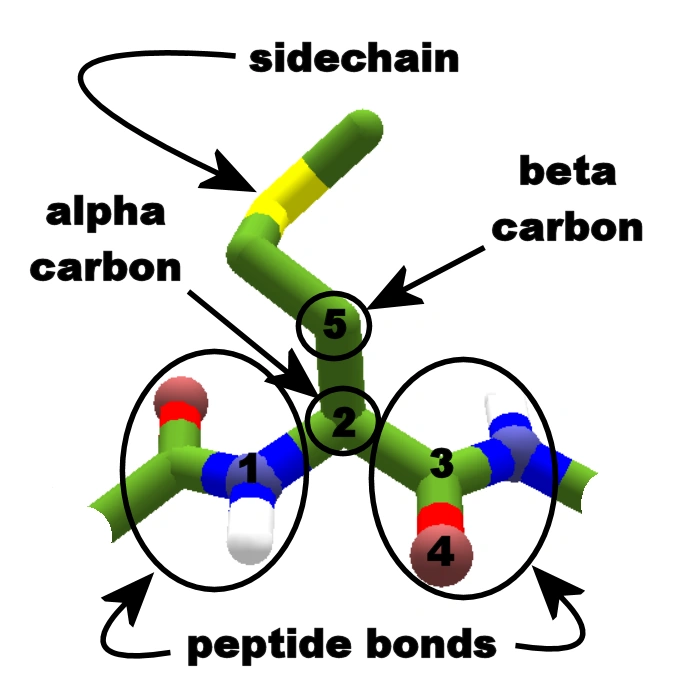
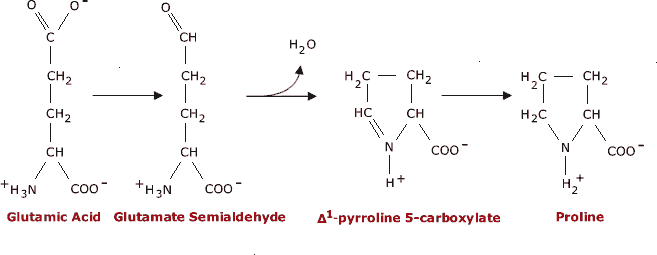
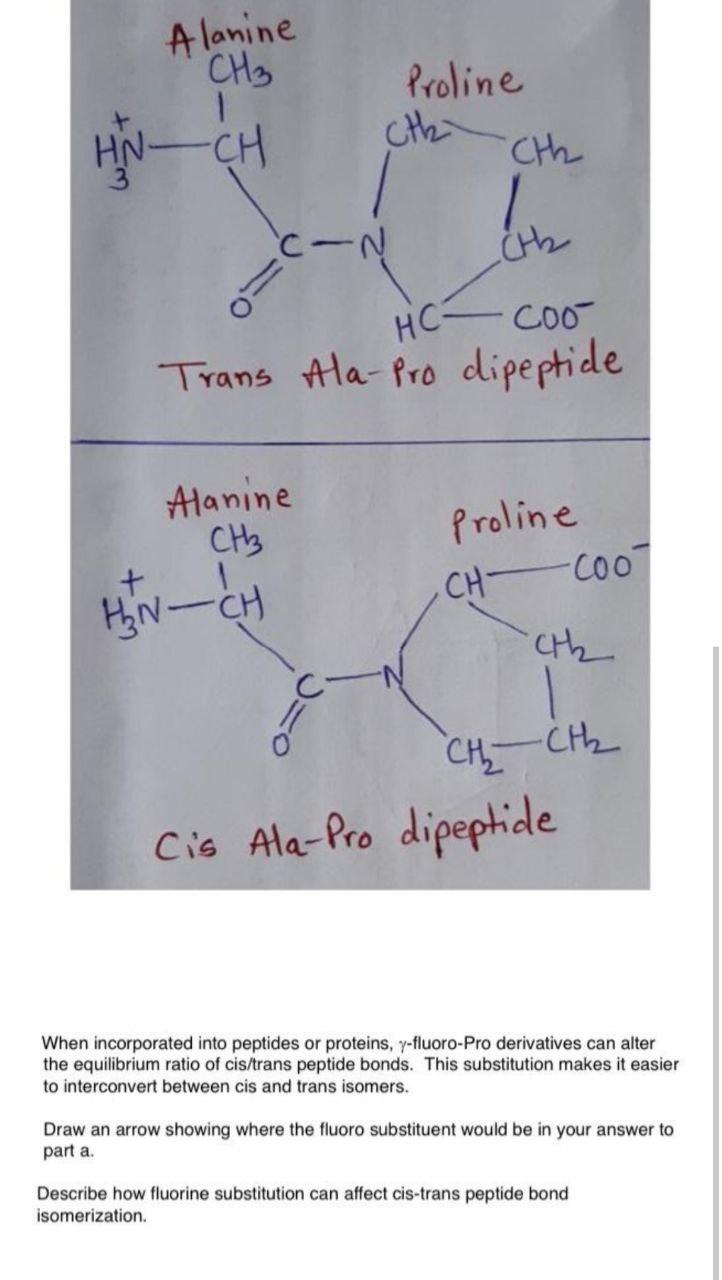
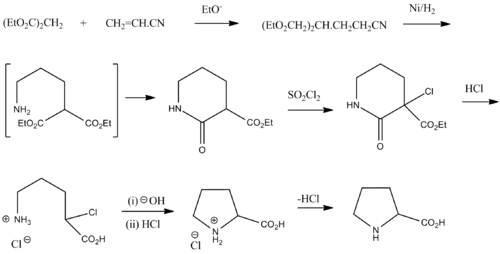
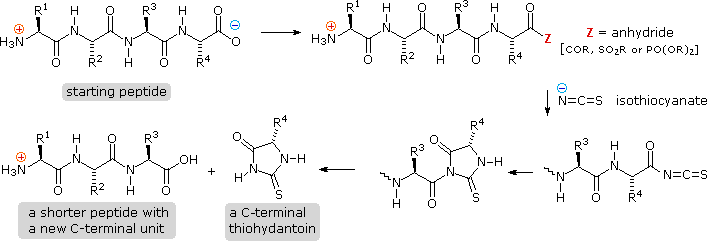
On the Cis to Trans Isomerization of Prolyl–Peptide Bonds under Tension | The Journal of Physical Chemistry B

Peptidyl-prolyl isomerisation. Cis/trans isomerisation of the peptide... | Download Scientific Diagram

Structural Basis of Proline-Proline Peptide Bond Specificity of the Metalloprotease Zmp1 Implicated in Motility of Clostridium difficile - ScienceDirect
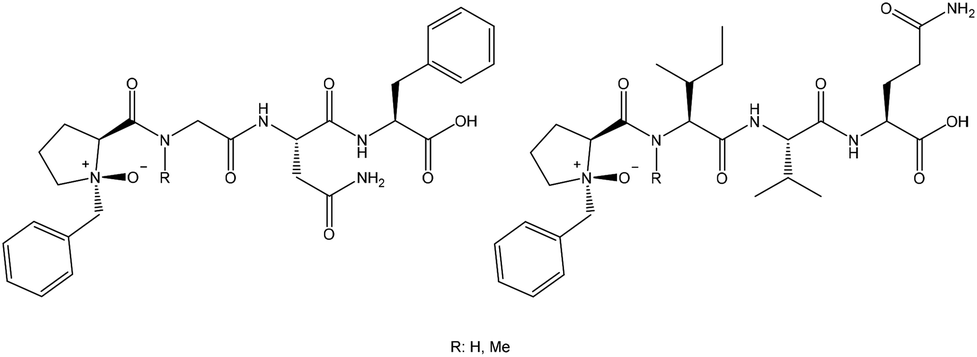
Proline N -oxides: modulators of the 3D conformation of linear peptides through “NO-turns” - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB00433G
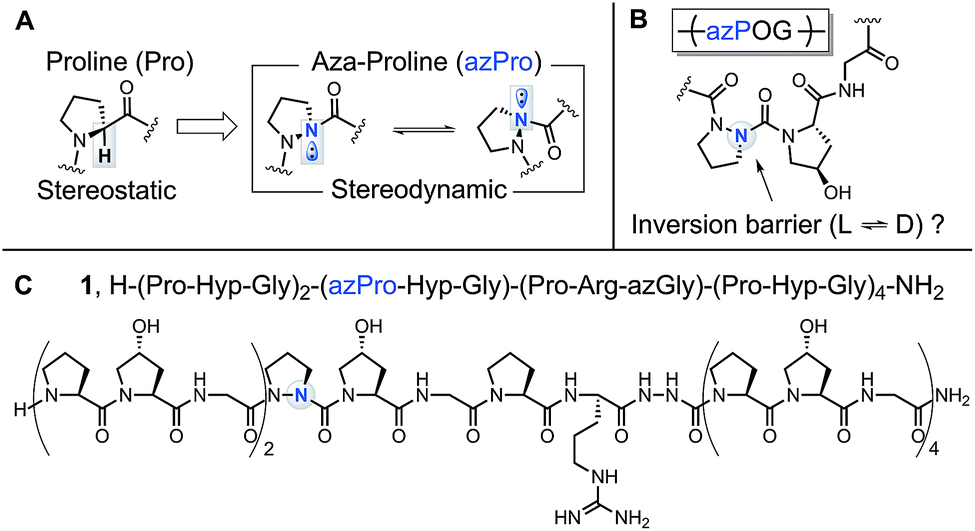
Aza-proline effectively mimics l -proline stereochemistry in triple helical collagen - Chemical Science (RSC Publishing) DOI:10.1039/C9SC02211B
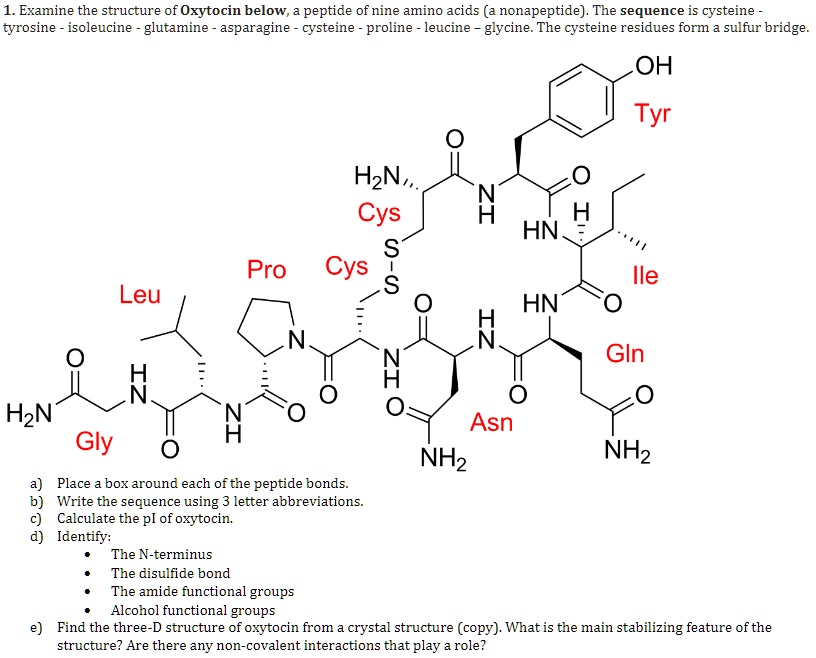
SOLVED: Examine the structure of Oxytocin below, peptide of nine amino acids (a nonapeptide) The sequence is cysteine tyrosine isoleucine glutamine asparagine cysteine proline leucine glycine The cysteine residues form sulfur bridge
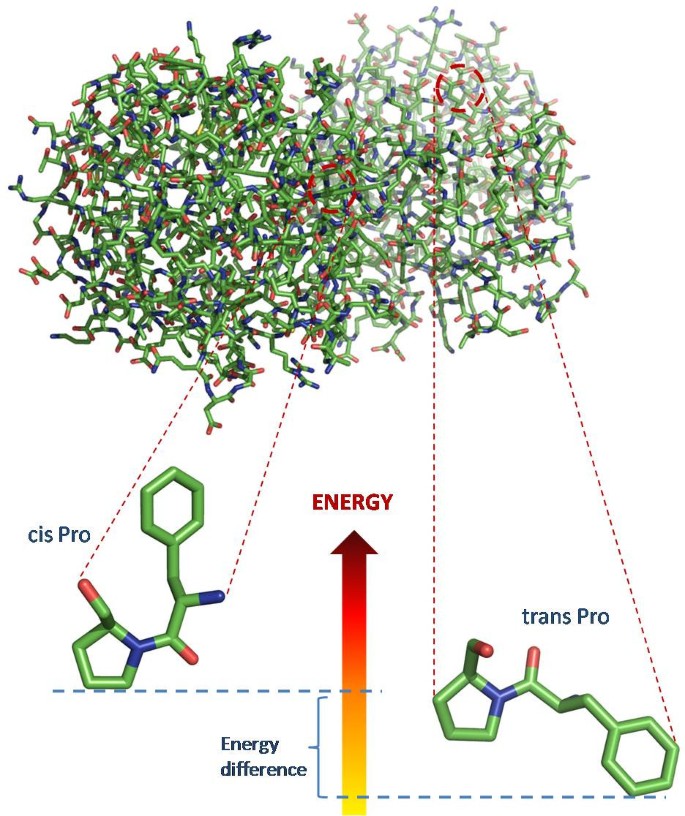
Detection of discriminative sequence patterns in the neighborhood of proline cis peptide bonds and their functional annotation | BMC Bioinformatics | Full Text